No Products in the Cart
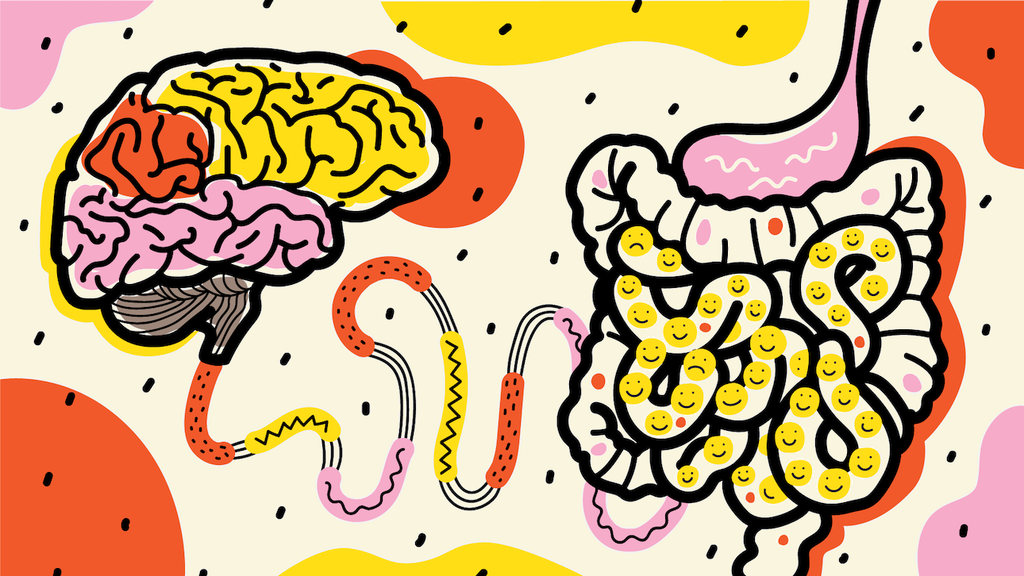
Have you ever had that "gut feeling" or "butterflies in the stomach"? This is something we experience because the gut and the brain are very closely connected. You could say that our emotions are felt in the gut, and when we feel stressed, it should not come as a surprise that it can lead to a range of gastrointestinal (GI) disorders and symptoms.
The enteric (aka gut) nervous system is often referred to as the "second brain" due to its complexity and the significant role it plays in regulating the digestive system. It contains over 100 million neurons, which is more than the number of neurons present in the spinal cord! The enterochromaffin cells of enteric nervous system are in direct contact with gut bacteria on one side and with the nerve endings on the other side. Although these cells account for less than 1% of intestinal epithelial cells, they produce and release up to 95% of the body's serotonin, a neurotransmitter that plays a crucial role in regulating mood (Xu et al., 2021; Terry and Margolis, 2017).
Stress and anxiety can disrupt the tenuous balance of the gut microbiome, leading to an overgrowth of harmful bacteria (Foster, Rinaman and Cryan, 2017). Stress response is perfectly normal and harmless in the short term, but serious issues can arise if we remain in a chronic state of stress. Prolonged stress has been linked to several disorders, such as obesity, heart disease, diabetes and even depression (Mariotti, 2015). In addition, it can worsen symptoms such as abdominal pain, bloating, diarrhoea, and constipation in multiple GI disorders including; Irritable Bowel Syndrome (IBS) and Inflammatory Bowel Diseases (IBD) (Foster, Rinaman and Cryan, 2017).
Studies have also shown that the gut microbiome can influence the brain and behaviour in various ways. The gut-brain axis is a communication network that acts as a bridge to connect the enteric nervous system to the central nervous system, allowing these two systems to communicate and influence each other (Mayer, Tillisch and Gupta, 2015). The gut microbiome plays a crucial role in this interaction, producing neurotransmitters and metabolites that can impact mood, cognition, and behaviour.
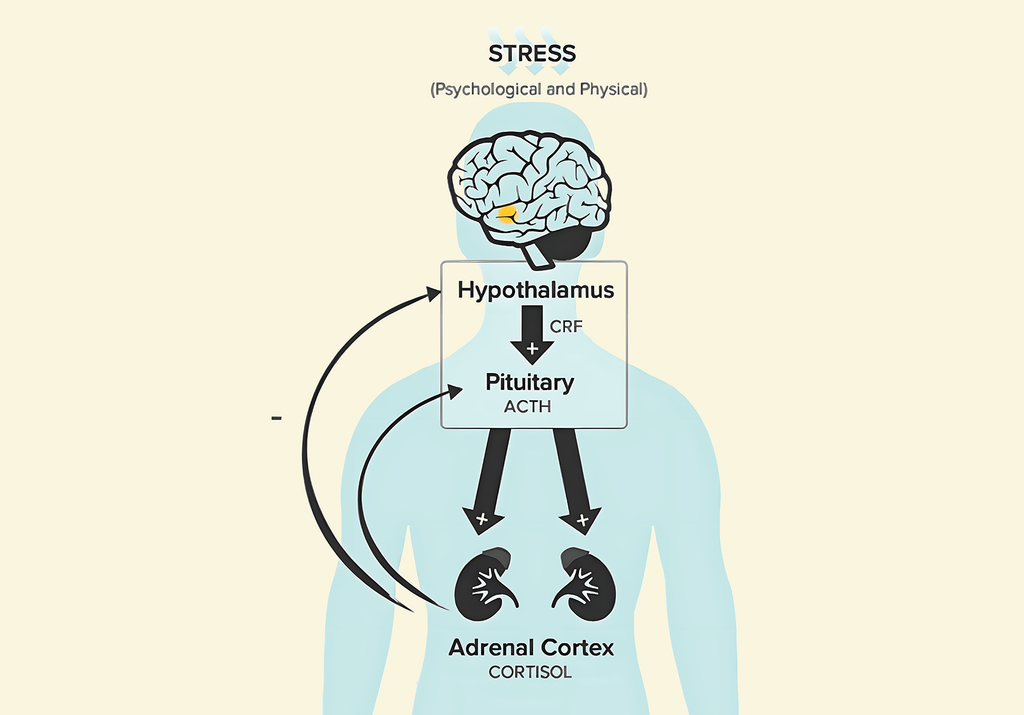
Our body's response to stress is regulated by the hypothalamic pituitary adrenal (HPA) axis complex system. When the brain perceives stress, it signals the hypothalamus to release certain hormones such as corticotropin-releasing factor (CRF), which then stimulates the pituitary gland to release adrenocorticotropic hormone (ACTH) (Smith and Vale, 2006).
This hormone then signals the adrenal glands located in the kidneys to release cortisol, which helps the body to cope with stress. However, chronic stress can lead to dysregulation of the HPA axis and cause an overproduction of cortisol, leading to negative health effects such as anxiety and depression (Smith and Vale, 2006).
The microbiome also affects the amygdala, another key stress-related brain region. The amygdala is a small almond-shaped structure that is responsible for regulating emotions and is connected to the gut through the vagus nerve. This connection allows stress and anxiety to impact gut function, leading to the symptoms mentioned above such as abdominal pain, bloating and changes in bowel movements (Foster, Rinaman and Cryan, 2017).
A leaky gut, on the other hand, as well as giving us an uncomfortable mental image refers to increased intestinal permeability, which has also been proposed to contribute to Major Depressive Disorder (Maes et al., 2009). This occurs when the epithelial barrier of the GI tract becomes compromised as a result of stress, which leads to an excessive release of gram-negative bacteria across the mucosal lining for access at the enteric nervous system (ENS) (Gareau et al., 2008).
Evidence for the importance of the role of microbiota in the regulation of stress-related changes in behaviour and brain function has primarily been researched in animal studies. An important discovery was made in 2004 when it has been observed that germ free mice, i.e. mice that have no bacteria in their gut, had an exaggerated HPA axis response to stress. The researchers also demonstrated that this response could be reversed by adding bacteria from the genus Bifidobacterium (Sudo et al., 2004).
Another study has also shown that changes in gastrointestinal motility, secretions, and blood flow can result from stressful events. They concluded from their analysis of the data that commensal microorganisms, or harmless bacteria living in our guts, can have an impact on the brain network that regulates how receptive we are to stress (Breit et al., 2018).
What we eat plays a major role in the regulation of our gut and stress response. For example, one study found that feeding mice a high-fat and sugar diet led to changes in gut microbiota composition and increased anxiety-like behaviours. Conversely, feeding mice a diet rich in fibre led to increased diversity of gut microbiota and reduced these behaviours (Bruce-Keller et al., 2015)
What should you eat to reduce the impact of stress?
A high-fibre diet can promote the growth of beneficial bacteria which produce short-chain fatty acids (SCFAs) such as butyrate and acetate. They have been shown to exert anti-inflammatory effect and modulates the production of neurotransmitters which can lower behaviours such as anxiety and depression (Gentile and Weir, 2018).
The type and amount of dietary protein can also influence the gut microbiota composition. Diets high in tryptophan, found in chicken, sunflower seeds, peanuts and pumpkin seeds, can stimulate the production of serotonin which regulates mood and anxiety levels (Linh, Guntoro and Qui, 2021).
The type of fat consumed also affects the composition of the gut. Diets high in saturated fats can lead to an imbalance of microbiota, known as dysbiosis, and increase inflammation (Rinninella et al., 2019). On the other hand, omega-3 fatty acids found in fish or algae and nuts have anti-inflammatory properties and can improve gut microbiota composition. Omega-3 supplementation has been associated with reduced anxiety-like behaviours in both animal and human studies.
Polyphenols are bioactive compounds found in plant-based foods such as fruits, vegetables, and whole grains. They can modulate the microbiota by promoting the growth of beneficial bacteria while inhibiting harmful ones (Etxeberria et al., 2015). They also have an antioxidant and anti-inflammatory properties that can influence anxiety levels. A diet rich in polyphenols has been linked to improved mental health and reduced anxiety-like behaviours.
Fermented foods like kvass, sauerkraut, kimchi, and kombucha are packed with live cultures. When you eat these foods, you're adding to the population of good bacteria in your gut. This can help keep your gut healthy by balancing the gut microbiome, thereby positively impacting your mood and mental health (Dinan and Cryan, 2017). Moreover, Lactobacilli bacteria present in fermented foods produce gamma-aminobutyric acid (GABA) during fermentation, which is a neurotransmitter that helps to reduce anxiety and promote relaxation. Additionally, the vitamins and minerals in fermented foods can bolster your overall health and wellbeing.
More research is needed to fully understand the mechanisms underlying these effects, but it is clear that our gut microbiome and diet can have an important role in managing our anxiety levels.